By Orville Williams
The public is being assured that, considering the sensitive nature of those involved, proper preparation will be done prior to accessing and administering Covid-19 vaccines to children aged five and 11.
Just last week, the US Centers for Disease Control and Prevention (CDC) gave the final seal of approval for the Pfizer and BioNTech vaccine to be rolled out for that age group in the United States.
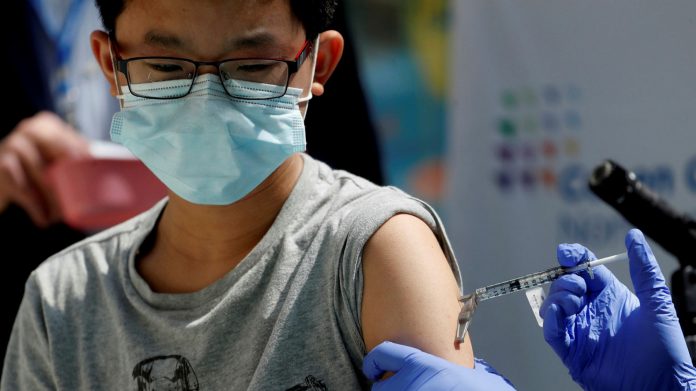
This is welcome news not only for the US but other countries that intend to vaccinate their younger population. Antigua and Barbuda is one of those countries, having voiced its intention early on, and repeatedly, to access the Pfizer vaccines and begin inoculating eligible children.
The process though, won’t be as straightforward as it is administering some of the Covid vaccines to adults, because these particular vaccines will have different doses, dilution requirements and storage conditions.
Along with these specifics, the fact that younger children – a vulnerable age group – are the ones receiving the jabs, the room for error is extremely narrow.
As far as Antigua and Barbuda is concerned, government says every effort will be made to meet all the necessary requirements before the rollout gets into gear.
“The government has been responsible at all levels with this particular matter…so, whatever technical and facility requirements there are, the government will absolutely put those in place before we take on board the challenge of deploying these vaccines in that environment – particularly to our tender little ones,” Information Minister Melford Nicholas said on Thursday.
The minister also noted that the government will continue leaning on the advice and assessment of the National Technical Working Group (NTWG) on Covid-19 vaccines, particularly toward this new development.
“We rely on the expertise and the professionals that are in that group to inform the process of the government [and] the Ministry of Health would have availed itself of all the prerequisites to be able to have vaccines being received, stored and deployed.
“So, yes, the government will remain responsible as it has been throughout the course of the vaccination process,” Nicholas said.
The government could also soon require more technical advice, in addition to more doses of the Pfizer shot for young children. On Thursday, during the handover ceremony of two field hospitals donated by the US, Prime Minister Gaston Browne requested the US also consider gifting more jabs along with Covid antiviral pills.
The latter, manufactured by US pharmaceutical company Merck, was approved for use last week by the UK’s Medicines and Healthcare products Regulatory Agency (MHRA).
A US Food and Drug Administration (FDA) advisory committee is scheduled to evaluate the safety and efficacy data of the pill on November 30 and decide whether to approve it for emergency use authorisation in the US.
